|
6/24/2023 0 Comments Copper electron configurationThe occupancy order for the orbitals has been shown as follows: In order to write the electron configuration, the occupancy order of the orbitals is needed to be determined first along with their principal quantum number. It implies that for any two different electrons, either one of the magnetic quantum number (ml), orbital angular momentum quantum number (l), spin magnetic quantum number (ms) or principal quantum number (n) needs to vary.  
According to Pauli-exclusion principle, any single electron can be described on the basis of four different sets of quantum number, which needs to vary in at least one. Aufbau’s principle states that electron of an atom, always occupy the orbital in the increasing order of energy, that is, ‘s’ orbital, followed by ‘p’ orbital, followed by ‘d’ orbital, followed by ‘f’ orbital. Hund’s rule states that an electron to double occupy a degenerate orbital, must first occupy them singly. There are three basic rules for electron configuration namely, Hund’s rule, Aufbau’s principle and Pauli-exclusion principle. Thus, the electron arrangement of an atom in accordance with the energy level at its minimum value, represents the ground state electron configuration of an atom. It is also referred to as zero-point energy. 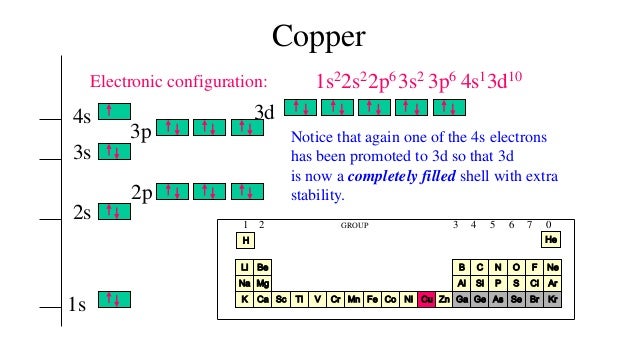
In quantum mechanics, ground state is defined as the state, which has the lowest possible energy. Give the actual ground-state electron configuration for copper (Cu) using the complete form.Įxpress the electron configuration using superscripts where appropriate.įor example, the configuration for Li would be entered as 1s^22s^1.Įlectronic configuration is the electron distribution of a particular atom in its molecular orbitals.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
